Ticker: GMAB
Stock exchange: Copenhagen
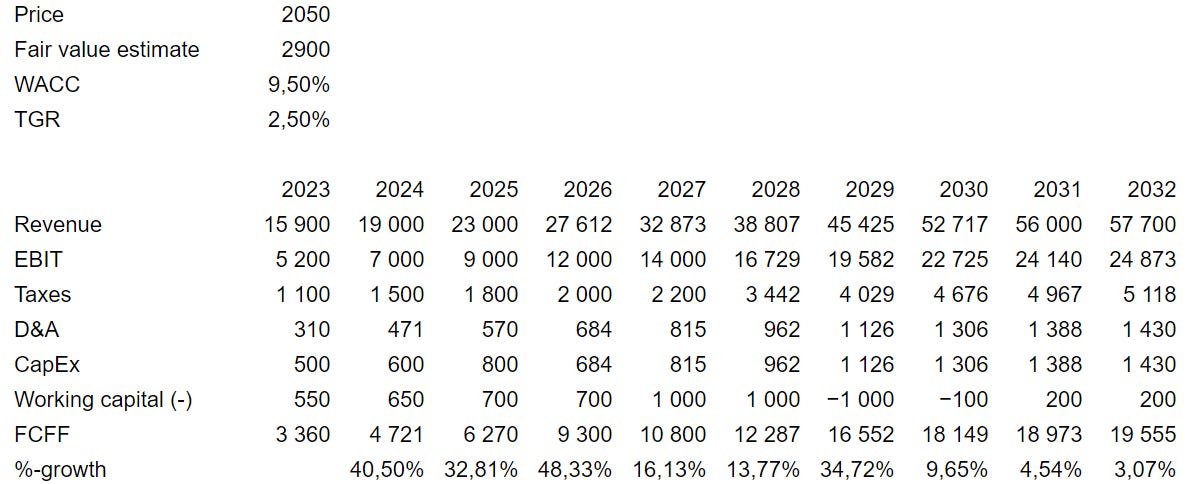
Price: 2050 DKK
Fair value estimate: 2900 DKK
Recommendation: Buy
Company Overview
Genmab (GMAB) is a biotechnology company founded in 1999 in Copenhagen, Denmark. The company specializes in the development of differentiated antibody therapeutics to fight against cancer and other serious diseases. GMAB’s headquarters is in Copenhagen, but it has also facilities in the Netherlands, USA, and Japan. It has around 2,000 employees and a market capitalization of close to $20B.
The anatomy of an exaggerated reaction
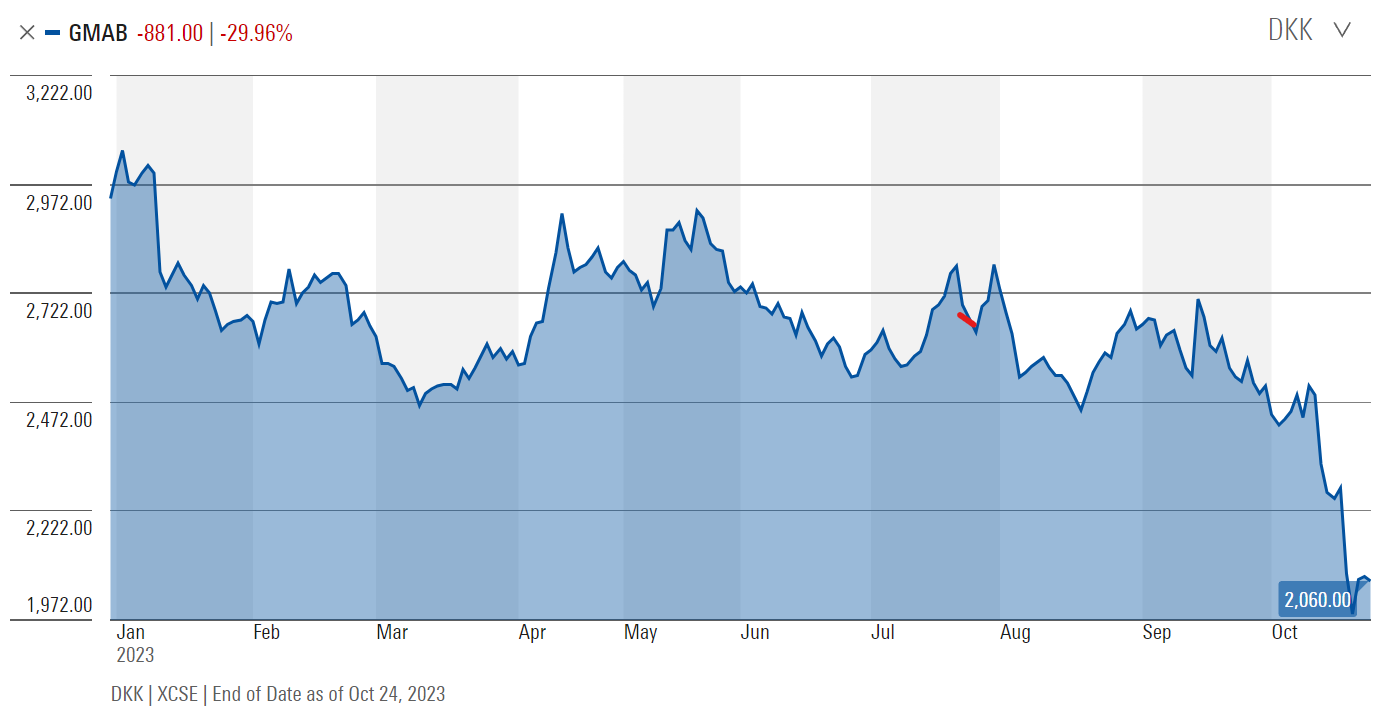
GMAB has experienced a significant decline of over 37% from its all-time high earlier this year, with most of this decrease occurring within the last month, as indicated in the chart provided below. This recent downturn can be attributed, in large part, to an announcement of lower-than-projected sales of the drug Darzalex, Genmab’s primary revenue contributor.
I think, however, that the market is putting too much emphasis on the sales GMAB generates today. In my view, the true potential of Genmab lies not in its present output, but rather in its future developmental trajectory. Therefore, in order to reach a realistic value of what the company is worth, we have to analyze the pipeline of new drugs, and from this vantage point form a balanced view of how sales and profit margins are going to look in the future.
Antibody therapeutics explained
Antibody technologies have become pivotal in modern cancer therapies. Antibodies are Y-shaped proteins that play a central role in immunity against pathogens (bacteria, viruses, and diseased cells). As we develop immunity, our bodies generate antibodies that bind to specific pathogen structures (known as antigens). Once bound, the antigens attract other parts of the immune system to eliminate the pathogen. In modern medicine, researchers have learned how to discover, create, and develop specific antibodies directed toward antigens associated with diseased cells for use in the treatment of cancer, autoimmune diseases, and other indications. It is in this realm that GMAB excels.
Genmab’s technology platforms
GMAB uses several types of technologies to create antibodies and has developed proprietary antibody technologies including the DuoBody, HexaBody, DuoHexaBody, and HexElect technology platforms. We will go through these platforms in more detail later. What is crucial to note at this point is GMAB's successful track record of creating therapeutic solutions leveraging these platforms. Hence, there is a high likelihood that the company will continue to introduce novel therapies derived from these technologies.
Moreover, it is noteworthy that GMAB harnesses its platform suites to construct extensive libraries of antibodies targeting specific antigens. These libraries are then licensed to other companies operating in the field of immunotherapy, underscoring GMAB's collaborative and innovative approach.
Before delving into the pipeline, it is prudent to examine the approved pharmaceuticals within GMAB's portfolio, providing a comprehensive foundation for our subsequent analysis.
Revenues today
GMAB expects revenues to be in the range of 15500 - 16500 million DKK in 2023. The revenue stream comes mainly from royalties of approved medicines that GMAB has co-developed with others as a minority partner. Here is a list of the approved medicines:
It is noteworthy that royalty income stipulates that GMAB is entitled to no more than 20% derived from the net sales of these medicines. As we navigate through the pipeline, our primary focus will be on clinical trials where GMAB stands to gain 50% or more from future sales. Anticipated ownership majority in these trials is poised to significantly elevate revenue streams in the coming decade. It must also be noted that GMAB will get 50 % of the total sales from the recently approved Tivdak.
Pipeline
These are the pharmaceuticals sorted under their respective technology platform:
DuoBody technology platform
The DuoBody technology platform is an innovative platform for the discovery and development of bispecific antibodies. Bispecific antibodies bind to two different epitopes (or “docking” sites) either on the same or on different targets (also known as dual-targeting). Dual-targeting may improve binding specificity and enhance therapeutic efficacy or bring two different cells together (for example, engaging a T cell to kill a tumor cell). GMAB uses the DuoBody technology platform to create its own bispecific antibody programs and the technology is also available for licensing. Genmab has numerous alliances for the DuoBody technology platform including commercial collaborations with AbbVie, Janssen, Novo Nordisk, BioNTech, and Immatics. Here is a list of ongoing trials under this umbrella platform:
The list doesn’t cover pre-clinical trials, license agreements, and collaborative research alliances.
HexaBody technology platform
The HexaBody technology platform is a platform that is designed to increase the potency of antibodies by strengthening the natural killing ability of antibodies while retaining regular structure and specificity. The technology allows for the creation of potent therapeutics by inducing antibody hexamer formation (clusters of six antibodies) after binding to their target antigen on the cell surface. Genmab is using the HexaBody technology platform for its own antibody programs and the technology is also available for licensing.
Two HexaBody-based investigational medicines are currently in clinical development. Genmab entered into an exclusive worldwide license and option agreement with Janssen to develop and commercialize HexaBody-CD38 (GEN3014), a next-generation CD38 monoclonal antibody-based investigational medicine. In 2022, Genmab and BioNTech expanded their global strategic collaboration to include co-development of monospecific antibody candidates leveraging the HexaBody technology. The first antibody in the clinic under this collaboration is HexaBody-CD27 (GEN1053/BNT313).
DuoHexaBody Technology Platform
The DuoHexaBody technology platform is a proprietary technology that combines the dual targeting of the DuoBody technology platform with the enhanced potency of the HexaBody technology platform, creating bispecific antibodies with target-mediated enhanced hexamerization.
GMAB has one investigational medicine created with the DuoHexaBody technology platform in the clinic, DuoHexaBody-CD37 (GEN3009). DuoHexaBody-CD37 is a bispecific antibody that targets two non-overlapping CD37 epitopes. It entered the clinic in 2020 and is currently being investigated in relapsed/refractory B-NHL, including potentially in combination with Epkinly.
HexElect Technology Platform
The HexElect antibody technology platform is Genmab’s newest proprietary antibody technology. This technology combines two HexaBody molecules designed to effectively and selectively hit only those cells that express both targets by making the activity of complexes of HexaBody molecules dependent on their binding to two different targets on the same cell. The HexElect technology platform maximizes efficacy while minimizing possible toxicity, potentially leading to more potent and safer investigational medicines. There are only pre-clinical trials going on under this umbrella for the time being.
Valuation
I am telling a bullish story about GMAB’s pipeline, foreseeing a substantial annual revenue growth of approximately 20% from 2025 to 2030. Additionally, I anticipate that the EBIT margin will eventually stabilize above 40%.
GMAB made a significant announcement on October 22nd, revealing that Tivdak has the potential to extend the life expectancy of women diagnosed with relapsed cervical cancer. This development is crucial given that patients with this diagnosis typically face a maximum life expectancy of 5 years. Consequently, I believe Tivdak will make a significant contribution to GMAB's overall revenue in the future.
Furthermore, Epkinly recently received fast-track approval in the US. I anticipate that this pharmaceutical will be as effective as Darzalex and may even be used in combination with it. Consequently, both Epkinly and Tivdak are poised to substantially enhance GMAB's sales figures in the upcoming years.
Looking ahead, GMAB has a robust pipeline of new pharmaceuticals in various stages of development, with several in Phase 2 and Phase 1. It is essential to recognize that research and development efforts continue beyond the approval of a medicine for a specific indication. Typically, a medicine demonstrates efficacy across a range of indications, either on its own or when combined with other therapeutic agents.
My DCF valuation follows below:
Conclusion
GMAB’s vision is to knock our socks off by 2030. I think it has a good chance of doing just that.
Disclaimer: Important Information for Retail Investors
The information provided in this blog is intended for educational and informational purposes only and should not be construed as financial advice or a recommendation to buy, sell, or hold any securities. Investing in individual stocks involves inherent risks, and past performance is not indicative of future results. Before making any investment decisions, it is crucial to conduct thorough research and consider seeking advice from qualified financial professionals.
The author of this blog is a retail investor and not a licensed financial advisor or registered investment professional. While the author strives to present accurate and up-to-date information, there is no guarantee that the content provided is accurate, complete, or current. Market conditions can change rapidly, and stock prices can be volatile.
All investments carry a degree of risk, including the potential loss of principal. Retail investors should carefully assess their risk tolerance and investment goals before making any investment decisions. Diversification is a key strategy to manage risk, and investing solely in individual stocks may expose investors to higher levels of risk compared to a diversified portfolio.
The author may have positions in the stocks mentioned in the blog. These positions may change at any time, and the author is under no obligation to update readers on such changes. It is recommended that readers do their own due diligence and consider seeking advice from qualified professionals before acting on any information presented in this blog.
Investors should be aware of the inherent limitations of the information available on the internet, including the potential for misinformation and bias. Always verify information from credible sources and cross-reference any data presented in this blog.
In accordance with prudent compliance, the author encourages readers to carefully review and understand the prospectuses, annual reports, financial statements, and other relevant information before making investment decisions. Retail investors should be aware of their own financial situation and consult with appropriate professionals to ensure that their investment choices align with their individual circumstances and goals.
By accessing and using this blog, readers acknowledge and agree that they are responsible for their own investment decisions and any outcomes that may result. The author and any related parties are not liable for any losses, damages, or actions arising from the use of the information provided in this blog.
In conclusion, investing in individual stocks carries risks that may not be suitable for all investors. Retail investors are advised to exercise caution, conduct thorough research, and consider seeking guidance from qualified financial professionals to make informed investment decisions.